Amnis Imagestream Channel Guide
Sample Preparation Guide
Fluorochrome Chart
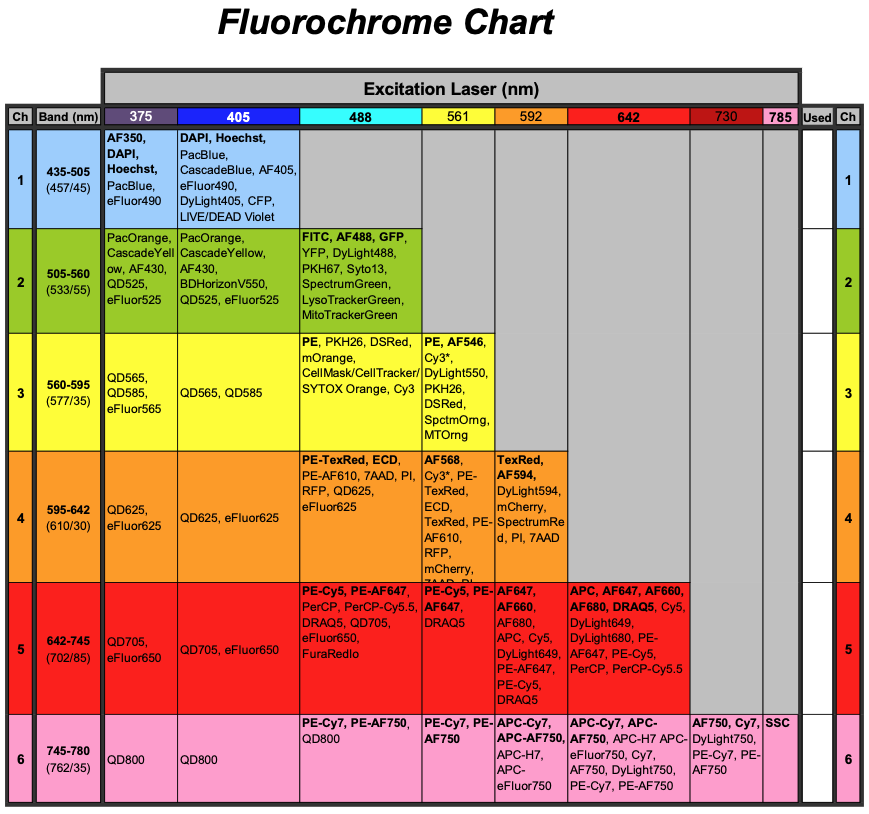
Recommended dyes (based on optimal excitation and detection channels) are in boldface.
*Many dyes will excite by more than one laser, and this can increase cross camera compensation.
**Channel bandpass may change depending on which lasers are on during an experiment. Values listed are assuming 405,488, and 642 laser excitation.
1 laser (488): ideal dyes are BF, AF488, PE, PE-TexRed, PE-Cy5, SSC
2 laser (488,642): ideal dyes are BF, AF488, PE, PE-TexRed, AF647, SSC
3 laser (405,488,642): ideal dyes are DAPI, AF488, PE, PE-TexRed, AF647, SSC/BF
Sample Preparation Guide
Experimental Design
The ImageStream system can quantify the intensity, specific location and distribution of signals within tens of thousands of cells per sample. The system can perform most flow cytometric assay, but the best applications take advantage of the technology’s imaging capabilities to discriminate subtle morphologic or signal distribution changes within individual cells and cell populations.
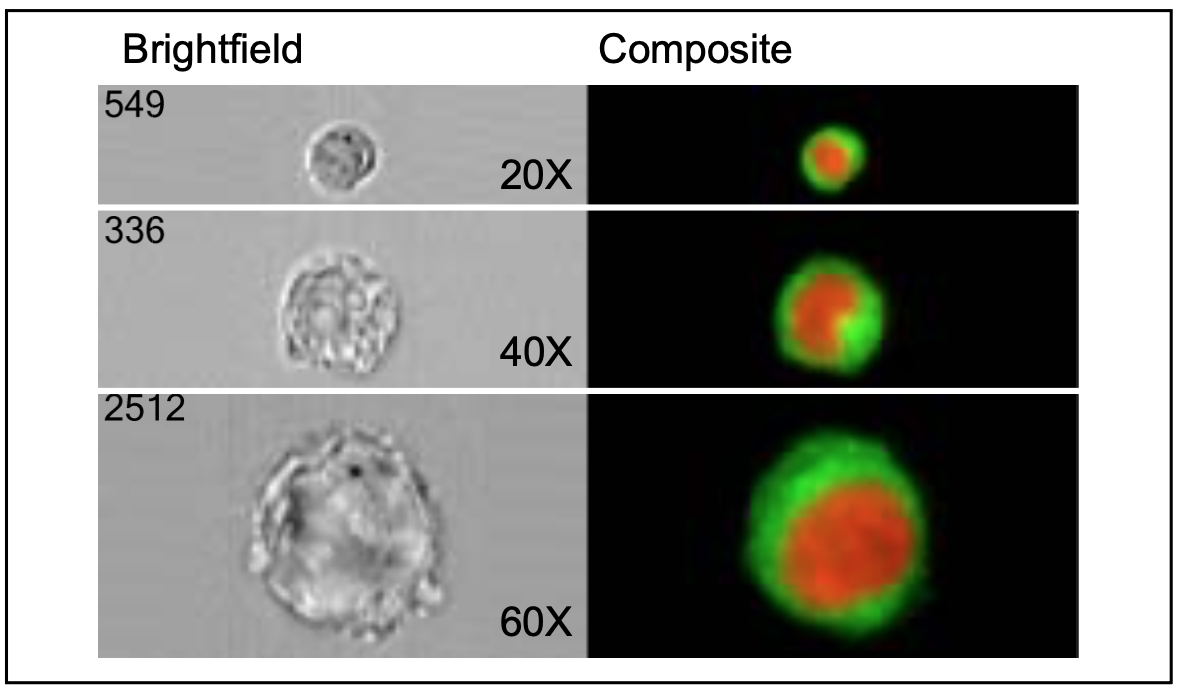
- Choice of Cell Type: The particle size should be less than 120um using 20x magnification, 60 um using
40x, and 40um using 60x. Images below are THP1 cells (~15um diameter) labeled with
FITC NFkB and Draq5.
-
Final Sample Concentration and Volume: At least 1 million cells in 50 µL (2x107 cells/ml) in PBS/2%FBS in a 1.5mL siliconized microcentrifuge tube.
-
Protocols: In general, any established labeling protocol used for flow cytometry will work with the ImageStream (see Current Protocols in Cytometry for general labeling techniques). Stain cells on ice in the presence of azide when possible to reduce non-specific capping of antibody. Use polypropylene tubes, preferable siliconized, to process samples.
-
Choice of Fluorochromes: Choose fluorochromes that are excited by the lasers in your ImageStream (405,488,642nm are most common). Use the chart at the top or look online for a spectra viewer that will help you plan which dyes will work the best.
-
Compensation: Have a sample of cells each labeled with a single-color for each fluorochrome used (i.e. FITC only cells, PE only cells, etc.).
-
Cell Aggregation: Minimize aggregation problems by straining the sample through a 70um nylon mesh strainer or by using an anti-clumping buffer such as EDTA or Accumax prior to fixation.
-
Fixation: If fixation is desired, thoroughly fix cells with 1% PFA on ice for 20 min.
-
Number of samples: No more than 30 total for feasibility experiments. Please limit the samples to the following: Positive and Negative biologic controls, compensation controls and experiment samples.
-
Brightness of Stain and Stain Balancing: Quantifying the location and distribution of signals in an image is a demanding task that requires optimized labeling. Below are a few suggestions to help design the experiment:
-
Try to achieve at least a full log shift in fluorescence, as measured by FACS.
-
Use the brightest dye for the antigen with the smallest copy number.
-
The brightness of probes can be independently controlled by changing the laser power. However, data quality is enhanced when the brightness levels of all probes excited off a single laser are balanced to within a log of each other. Probe balancing avoids the saturation of bright stains when they are combined with dim stains in the same sample.
-
-
Shipping recommendations:
-
Include the data acquisition form and data that verifies the experiment worked.
-
Samples must be fixed and non-pathogenic.
-
Wrap sample tubes in ParaFilm.
-
Insulate by placing in 15ml conical rack with Styrofoam lid. Pack in Styrofoam lined shipping box with refrigerant packs and paper. For winter delivery, use room temperature packs to prevent freezing of sample. For summer delivery, use frozen packs.
-
Label outside of box "Do Not Freeze."
-
Email the package tracking number to your Amnis contact person.
-
-
International Shipments: To ship non-viable (fixed) material internationally to Amnis, a written statement for US Customs and Border Protection, Department of Homeland Security must accompany the shipment. This written statement must:
-
Be addressed to: U.S. Customs and Border Protection, Department of Homeland Security.
-
Be an original copy on institutional letterhead signed by the laboratory worker responsible for preparing the samples.
-
Identify the material and name of the species from which the material was derived.
-
State that the animals from which the material was derived:
-
have not been exposed to, or inoculated with, any livestock or poultry disease agents exotic to the United States, and
-
did not originate from a facility where work with exotic disease agents affecting livestock or avian species is conducted
-
-
State that the material is non-viable.
-
Be placed in an envelope addressed to "U.S. Customs and Border Protection, Department of Homeland Security" and attached to the outside of the shipping box.
-
Shipping Address:
Cytek Bioscience/Amnis
645 Elliott Ave. W., Suite 100
Seattle, WA 98119
